In Quest of Events Leading to Pigmentary Disorders
in Several Mutants of the Budgerigar
[Melopsittacus undulatus]; a Light- and Electronmicroscopical Survey
By: Inte Onsman, Research coordinator
MUTAVI
Research & Advice Group, The Netherlands
Page revised december 29 2003
The diversity of colormutations in birds results in a large part from dysfunctioning of the
pigmentary system. Melanin is responsible for at least six major basic colors in bird feathers:
black, grey, cinnamon brown, redbrown, chocolate brown and white (absence of melanin).
Several functions have been proposed for pigmentation from which the most important is
camouflage. In the pigmentary system the melanocyte is the site of pigment production,
melanin is formed within melanosomes, membrane bound organelles located in the cytoplasm
of the melanocyte.
Melanocytes are found predominantly in the basal layer of the skin as well as in the feather-follicles. In addition, pigment cells reside in the retina and uveal tract of the eye.
Deposition of melanin in the retina of the eye occurs primarily dural fetal development and, 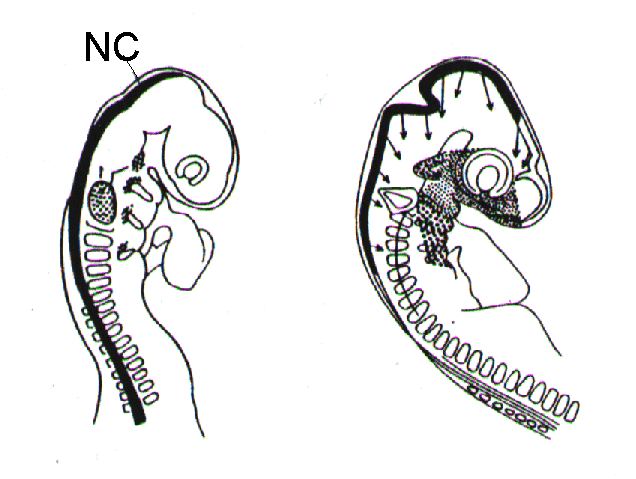 continues in birds for several days after hatching.
continues in birds for several days after hatching.
The vast majority of pigment cells are derived from precursor cells in the embryonic neural
crest (fig.). One exception is the pigmented layer of the retina (from which during fetal
development eventually the inner pigmented layer of the iris is separated) -its cells originate
from the optic cup of the forebrain [2, 9, 13].
Melanosomes contain:
(1) structural matrix proteins forming a lamellar matrix within the melanosome upon which
melanin is deposited.
(2) the enzyme tyrosinase, which catalizes the initial step in the melanin biosynthetic
pathway, and
(3) several proteins of unknown structure and function.
Four representative stages in melanosome development have been described;
1. Stage I melanosomes are spherical and may contain filaments with a distinct periodicity.
2. Stage II melanosomes are oval in shape and have numerous parallel longitudial filaments
with no melanin deposition.
3. Stage III melanosomes have a high level of tyrosinase activity and their internal structure is
partially obscured by the deposition of melanin pigment.
4. In stage IV melanosomes there are very low levels of tyrosinase activity and melanin is
deposited throughout the structure, rendering it electron-opaque.
Normal bird melanocytes have long dendrites to disperse their melanosomes into growing
feather barbs. As melanosomes pass from the perinuclear area of the melanocyte to these
dendrites, they progress from stage I to stage IV melanosomes.
In the melanin biosynthetic pathway, tyrosinase plays an important regulatory role -its level
of activity usually correlates with the degree of pigment production. In birds affected with
tyrosinase negative (ty-neg) albinism, such as in NSL inos, this correlation is most evident.
This type of albinism does not affect the morphology of the melanocyte or the
premelanosome.
Two types of melanin play a major role in aviculture: eumelanin and phaeomelanin. Only one
major form of melanin exist in psittacine birds; eumelanin. The latter is a brown to black
pigment, unsoluble in alkali. Phaeomelanin, on the other hand, is a red to foxbrown pigment,
fully soluble in alkali found in finch like birds and poultry.
Pigmentary disorders
Albinism is an inherited congenital disorder in which there is a generalized decrease (fallows)
or absence (inos) of pigment in the eyes, skin and feathers of mutant birds [1, 4, 7, 8, 12, 14,
15, 16, 18, 19, 22, 23, 24, 26, 27, 31].
Although all areas have a normal number of melanocytes, the ability of those cells to produce
melanin is either reduced or absent. Tyrosinase negative (Ty-neg) albinism is characterized
by a complete lack of tyrosinase activity (NSL inos) or a decreased activity (bronze fallow in
the Budgerigar?) [4, 8, 18, 21]. In birds with tyrosinase positive (Ty-pos) albinism (sex-linked inos) more than twice the amount of standard levels of tyrosinase activity has been
found [3] suggesting there may be an abnormality in the regulation of more distal steps in the
melanin pathway [17, 32]. In addition the author would like to hypothesize that these very
high tyrosinase levels might be related to the increased size and amount of brown eumelanin
granules seen in the cortex of the wingcoverts of cinnamon-inos which is most striking in
Budgerigars [22, 23, 28, 29].
All types of albinism in birds, whether they inherit autosomal or sex-linked, are recessive
traits [1, 4, 7, 12, 14, 15, 16, 18, 19, 21, 22, 24, 26, 30, 31].
The primary clinical findings in albinism include;
dilution of the color of the feathers, skin and eyes (iris and fundus), however, no decreased
visual acuity is seen in Budgerigars [32] in contrast with sex-linked ino Redrumped- and Indian Ringnecked parrots.
Classification
Pearson, Nettleship, and Usher (1911) proposed the following classification of different
degrees of albinism:
1. Total: complete absence of melanin.
2. Incomplete: eyes, skin, or feathers affected, but not all three.
3. Imperfect: partial inhibition of pigment formation, but not total absence of pigment in any.
4. Partial: complete or partial inhibition of pigment formation within localized areas, as in
piebald albinism.
Examination
Several mutant feathers were investigated on the ultrastructural level. My laboratory work-up
included:
1. an examination of presumed abnormal eumelanosomes in wingcoverts taken from sex-linked inos and cinnamon-inos.
2. the same examination was carried out on wingcoverts from lightgreen cinnamons, bronze
fallows, spangles and the wild-type.
The results of the examination mentioned at point two above, will be discussed in the last part
of this article.
Sex-linked inos and cinnamon-inos
Barbs of fully developed wingcoverts taken from sex-linked inos and cinnamon-inos, contain
small amounts of morphologically abnormal eumelanosomes. In cinnamon-inos the amount
of these irregular and severely abnormal granules is 2.5 times higher than observed in sex-linked inos.
It might be possible that the interaction between the sex-linked ino and cinnamon loci result
in an increase of size and amount of dispersed abnormal eumelanosomes in the cortex of the
wingcoverts of sex-linked cinnamon-inos. This event leads to a very obvious brown laced
wing pattern in Budgerigars from which the name "lacewing" erroneously was adopted to use in other psittacine species. The presence of
these organelles also cause the faint ghost pattern seen on the back of the head and
wingcoverts of sex-linked ino Budgerigars. This phenomenon has also been reported in
turkeys, fowls and quails [11, 12, 16, 19, 31].
In other psittacine birds, cinnamon-ino combinations have been developed but unfortunately
the undulated "laced" wingpattern, which is so characteristic in Budgerigars, is completely
absent in these species. For the sake of clarity and to avoid confusion, the author would like
to propose to abandon the use of the name "lacewing" in other psittacine species. The term
"cinnamon-ino" is much more appropriate because we have more than overwhelming
evidence that these birds are in fact the result of crossing-overs between these two sex-linked
loci. Thus, they are genuine cinnamon-inos and do not represent a separate locus or allele as
several authors have postulated in the past [23, 28].
In a paper published in 1987, avian albinos were discussed [3]. It was demonstrated on the
ultrastructural level (electron microscope) that only morphologically abnormal melanosomes
within sex-linked ino melanocytes are positive for tyrosinase activity, whereas numerous
morphologically normal premelanosomes are devoid of the enzyme.
Possible mechanisms for the selective accumulation of tyrosinase into abnormal
premelanosomes of sex-linked inos, can be devided into defects affecting premelanosomal
(matrix) synthesis or the tyrosinase transporting system [17, 33, 34].
Since no normal eumelanosomes were found in the cortex of feather barbs examined during
this investigation, my results suggest that there is also an exclusive dispersion of
morphologically abnormal eumelanosomes into featherbarbs of sex-linked inos and
cinnamon-inos. Possible mechanisms leading to this event are, as far as I am aware, still
obscure.
Cinnamon versus wild-type
An examination of cross sections at a magnification of 6900x shows that the shape of the
eumelanosomes in the outher cortex of cinnamon wingcoverts is spherical, and of a diameter
equal to about half to two thirds of the length of those seen in the wild-type. On the
other hand, also long oval shaped granules are to be found whithin densely packed groups in
more central parts of the cortex.
The quantity of granules in the cortex of the "cinnamon green" barbs examined, is
approximately the same, but in some specimens about 10% less than in corresponding regions
of the wild-type.
Under the lightmicroscope the color of the cinnamon eumelanosomes is indeed of a brown
shade. Examinations of eye tissues of cinnamons also show brown eumelanin in the
pigmented layer of the retina as well as in the choroid.
Bronze fallow
The phenotype of the bronze fallow in the Budgerigar is mainly characterized by greyish
brown colored wingcoverts. In order to elucidate the nature of the "greyish" brown, cross sections were examined at the ultrastructural level. In one cross section of a barb of a
wingcovert feather, I was fortunate to find a pigment cluster within a cortical cavity as seen in one of the photographs. The quality of
the eumelanin in this cluster is very irregular and poor. This can also be observed in detail in the cortex.
My assumption that the bronze fallow is allelic with the NSL ino is based on the fact that
deletions or point mutations in the tyrosinase gene (a - locus) give rise to loss or reduction of
enzyme activity, and thus to (almost) complete or partial loss of all pigment.
Decreased or poor tyrosinase activity could have led to the bronze fallow phenotype making it a serious candidate to be
allelic with the NSL ino in the Budgerigar, however, since NSL inos are very rare, we
have not been able to perform testmatings and so we are still waiting for evidence.
It is most likely that the mixture of "normal", poor and very poor quality eumelanosomes,
causes the typical "greyish" brown appearance on the wingcoverts of bronze fallows.
The spangle
In spangles a narrow zone of densely packed spherical eumelanosomes has been found
predominantly just beneath the surface of the cortex. The size of these granules varies
sometimes from very small to "normal". Large eumelanosomes are situated in the medullary
region enclosed by a spongy zone. Some of these granules show severe damage (arrows).
Whether this is due to the cutting of the knife or the action of some (bio)chemical agent is
unclear to me at this moment.
In the barbs of breast feathers of a DF spangle, many macro
melanin globules (fka macro- melanosomes) have been found [20]. These have also
been found in cross sections of wingcoverts studied under the lightmicroscope.
The Sp mutation is a semi-dominant trait that causes clumping, a typical irregular
distribution, and reduced numbers of melanosomes, obviously followed by premature death of
follicular melanocytes.
Since DF mutant birds show the real action of a mutant gene rather than a SF mutant bird,
these results suggest that some kind of melanocyte selfdestructing mechanism could be
responsible for the spangle phenotype in Budgerigars. Also abnormal phacocytical processes
affecting melanocytes cannot be ruled out [25]. However, more research on the biochemical
level might be necessary to elucidate the nature of this mutation.
Photographs of cross sections
(for best results use a colorprinter and glossy paper)
Pigment metabolism - Four stages of melanosome development -
Sex-linked albinism -
Lacewing -
Cinnamon - Cinnamon (detail) - Wild-type -
Bronze fallow -
Bronze fallow (detail) - Bronze fallow (another detail) -
Spangle - Spangle (detail)
SF Spangle - DF Spangle -
Pigment epithelium (cross section)
- Pigment epithelium (longitudial section) -
Developing choroidal eumelanosomes -
Normal choroidal eumelanosomes -
Abnormal choroidal eumelanosomes - Eye color
|
Consulted and cited literature:
[1] Bitgood J.J., Smyth J.R., (1991)
Albinism in White Leghorn Chickens
Poultry Science Vol.70; p.p.1861-1863
[2] Boichuk N.V., Chelishev Y.A., (1987)
Role of the Neural Crest in Development of the Avian Eye
Arkh.Anat.Gistol Embriol.93(10); p.p.62-66
[3] Boissy R.E., Moellmann G.E., Halaban R., (1987)
Tyrosinase and Acid Phosphatase Activities in Melanocytes from Avian
Albinos
Journ.Invest.Derm. Vol.88 no.3; p.p.292-300
[4] Brumbaugh J.A., Barger T.W., Oetting W.S., (1983)
A "new" Allele at the C Pigment Locus in the Fowl
Journal of Heredity Vol.74; p.p.331-336
[5] Brumbaugh J.A., Oetting W.S., (1986)
What Can We Learn from Chick Embryo Melanocytes?
Bioscience Vol.36 no.6; p.p.381-387
[6] Carr J.G., (1957)
Internal Structure of Avian Melanin Granules: An Electron Microscope Study
Quarterly Journ. of Micr. Sc. Vol.98; p.p.159-162
[7] Cole R.K., Jeffers T.K., (1963)
Allelism of Silver, Gold, and Imperfect Albinism in the Fowl
Nature Vol.200; p.p.1238-1239
[8] Einig A., (1993)
Non Sex-Linked Lutinos
Budgerigar World Issue 128; p.p.22
[9] Foulks J.G., (1943)
An Analysis of the Source of Melanophores in Regenerating Feathers
Physiological Zoology; p.p.351-380
[10]Francois J., Rabaey M., Lagasse A., (1963)
Electron Microscopic Observations on Choroid, Pigment Epithelium and Pecten
of the Developing Chick in Relation to Melanin Synthesis
Ophthalmologica Vol.146; p.p.415-431
[11]Frens D.B., (1991)
The Causes of Colour in Ino's
Bugerigar World Vol.12; p.p.10-11
[12]Hutt F.B., Mueller C.D., (1942)
Sex-Linked Albinism in the Turkey
Journ.of Heredity Vol.33; p.p.69-77
[13]Ide C., (1972)
The Development of Melanosomes in the Pigment Epithelium of the Chick
Embryo
Zeitschr.fur Zellforsch.Vol.131; p.p.171-186
[14]Ilhenny Mc.E.A., (1940)
Albinism in Mockingbirds
Journal of Heredity Vol.31; p.p.433-438
[15]Kokemuller K., (1935)
Geschlechtsgebundene Vererbung bei der Totalalbinotischen Aberration des
Melopsittacus Undulatus [Shaw]
Zeitschr.Ind.Abst.Vererb. Vol.21; p.p.299-302
[16]Lauber J.K., (1963)
Sex-Linked Albinism in the Japanese Quail
Science Vol.146; p.p.948-950
[17]Maul G.G., Brumbaugh J.A., (1971)
On the Possible Function of Coated Vesicles in Melanogenesis of the Regenerating Fowl Feather
Journal of Cell Biology Vol.48: p.p. 41-48
[18]Moore K., (1990)
The Reappearance of the Non Sex-Linked Red Eye
Budgerigar World issue 89, no.1; p.p.18-19
[19]Mueller C.D., Hutt F.B., (1941)
Genetics of the Fowl- Sex-linked imperfect albinism
Journal of Heredity Vol.32; p.p.71-80
[20]Nakagawa H., Hory Y., Sato S., (1984)
The Nature and Origin of the Melanin Macroglobule.
Journal of Invest.Derm. Vol.83 no.2; p.p.134-139
[21]Oetting W.S., Churchilla A.M., Yamamoto H., (1985)
C Pigment Locus Mutants of the Fowl Produce Enzymatically Inactive
Tyrosinase-like Molecules
Journ.Exp.Zool. Vol.235; p.p.237-245
[22]Onsman I., (1993)
The Lacewing: An Enigma in Budgerigar Breeding
The Budgerigar Journal (March Issue); p.p.9-12
[23]Putt C., (1993)
Lacewing Budgerigar a Mutation in its Own Right
Cage & Aviary Birds no.2; p.p.7
[24]Sage B.L., (1962)
Albinism and Melanism in Birds
British Birds Vol.55 no.6; p.p.201-225
[25]Saito N., Seiji M., (1973)
Epidermal Lysosome and the Degradation of Melanosomes
Acta Derm. Suppl. Vol.73; p.p.69-74
[26]Silversides F.G., Merat P., (1991)
Homology of the s+ Locus in the Chicken with Al+ in the Japanese Quail
Journal of Heredity Vol.82 no.3; p.p.245-247
[27]Speier H., Speier G., (1988)
Histologische Untersuchungen der Augen von Albinozebrafinken
AZN no.3; p.p.154-156
[28]Taylor T.G., Warner C., (1961)
Genetics for Budgerigar Breeders
ILIFFE BOOKS LTD, LONDON
[29]Taylor T. G., Warner C., (1986)
Genetics for Budgerigar Breeders
Publ.The Budgerigar Society; p.p.135
[30]Warren D.C., (1933)
Inheritance of Albinism in the Domestic Fowl
Journal of Heredity Vol.24; p.p.379-383
[31]Werret W.F., Candy A.J., King J.O.L., (1959)
Semi-Albino: A Third Sex-Linked Allelomorph of Silver and Gold in the Fowl
Nature Vol.184; p.p.480-482
[32]Wilken H, (1998)
Microscopical -Anatomical Investigations of the Eyes of "Albinotic" and Normal
Pigmented Color Varieties of the Budgerigar [Melopsittacus undulatus]
Thesis (School of Veterinary Medicine Hannover, bronzey)
[33]Yamamoto H., Ito K., Ishiguro S., (1987)
Gene Controlling a Differentiation Step in the Quail Melanocyte
Dev.Genetics Vol.8; p.p.179-185
[34]Zimmerman J., (1982)
Four New Proteins of the Eumelanosome Matrix of the Chick Pigment
Epithelium
Journ.of Exp.Zool. Vol.219; p.p.1-6
©Inte Onsman
MUTAVI Research & Advice Group
Back Home
 continues in birds for several days after hatching.
continues in birds for several days after hatching.